Come join us at the European Geosciences Union (EGU) General Assembly (Booth #10)
May 3-8, 2026 - Vienna, Austria
δ13C Compound Specific Isotope Analysis (CSIA) of gas phase hydrocarbons and atmospheric gas has been used to explain microbial, thermogenic or geogenic activity of gas, and provides a more robust understanding of the source of carbon than concentration alone. Applications that have used isotopic analysis include:
SGS Beta offers a specialized analysis of the following gases:
Select gases for chromatography isolation prior to combustion include:
SGS Beta uses international secondary reference materials (RMs) for the determination of the compound-specific δ13C values of hydrocarbon gases. The RMs are sourced from the United States Geological Survey (USGS), which provides a suite of methane, ethane, and propane as standard gases (HCG-1, HCG-2, and HCG-3), in addition to NIST SRM 4990C oxalic acid standards.
Below is an example of how δ13C analysis is applied in real-world environmental monitoring.
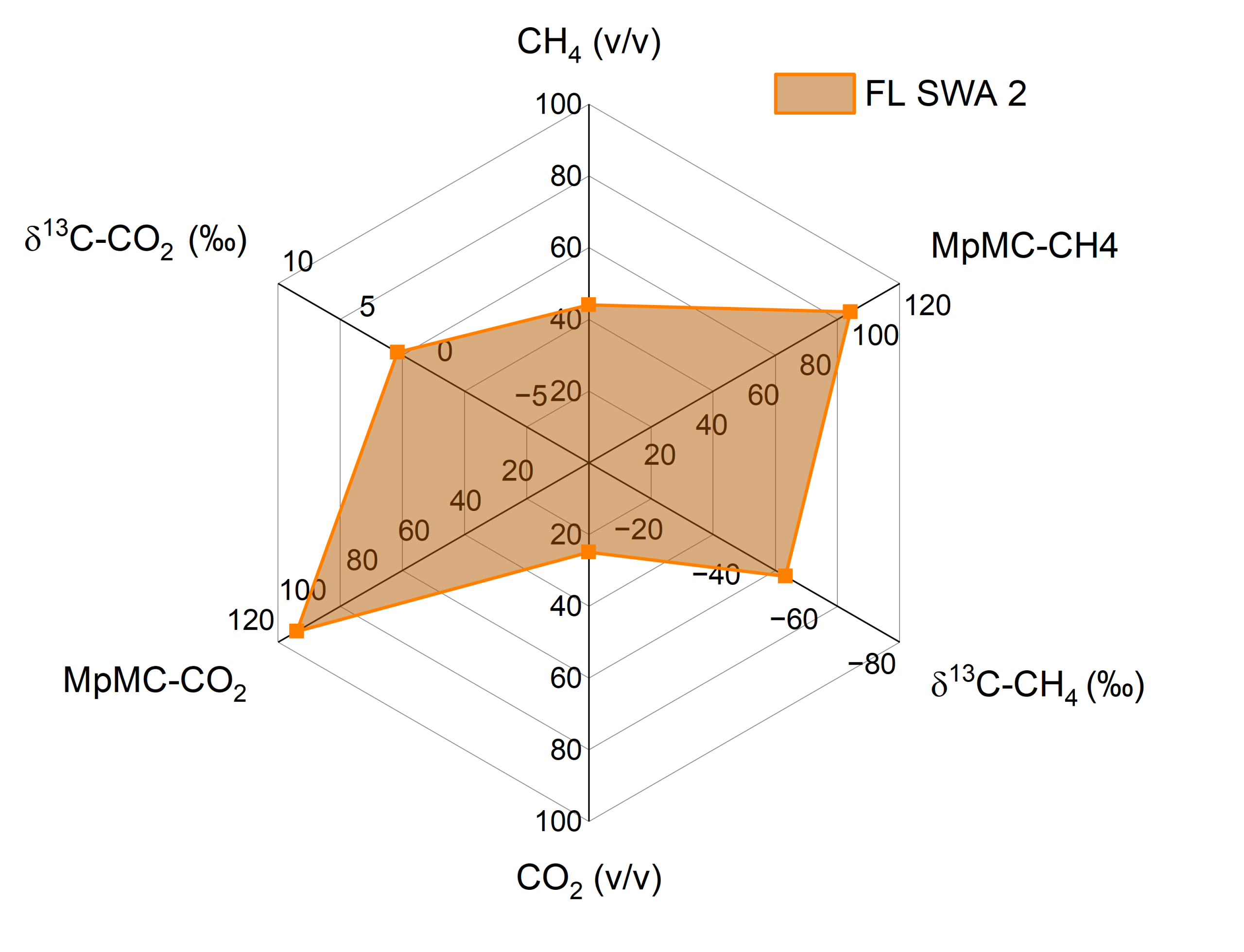
Figure 1: Class III Flare 1 landfill analyses of six parameters measured at SGS Beta (R&D Department)
δ13C analysis of CH4 and CO2 in soil gas and landfills has become an essential tool for distinguishing between biogenic and geogenic carbon sources. Methane in soils may originate from microbial methanogenesis, thermogenic leakage of fossil fuels, or even abiotic reactions. Each of these processes undergo 13/12C isotopic fractionation to produce δ13C signatures. For example, microbial pathways have shown in the scientific literature ¹³C-depleted values of methane from –50 to –110‰, thermogenic gas is moderately depleted (–30 to –50‰), while abiotic or mantle-derived methane can be comparatively enriched (–5 to –30‰). By measuring δ13C alongside depth profiles, methane sourcing can be inferred by actively forming in situ under anaerobic conditions, or migrated fossil gas contaminating shallow soil layers. The δ13C of the CO2 fraction provides complementary insight into carbon cycling and potential contamination when compared to the CH4 fraction. Soil-respired CO2 from vegetation is typically more negative in C3 ecosystems (–22 to –37‰) and less depleted in C4 systems (–9 to –15‰). Deviations from these ranges such as enrichment toward 0‰ may indicate carbonate dissolution or thermogenic degassing rather than root or microbial respiration. δ13C enrichment of the CO2 fraction from landfill gas has also been observed from –10 to +15‰.
δ13C analysis of ethane (C2H6) and propane (C3H8) provides critical context for interpreting gas origins alongside methane. Since these higher-chain hydrocarbons are rarely produced through microbial pathways, their presence and isotopic composition serve as key indicators of thermogenic or abiotic processes. In environmental and energy applications, enriched δ13C values in ethane and propane can help distinguish fossil fuel leakage or fracking impacts from purely microbial methanogenesis, while relative shifts among CH4, C2H6, and C3H8 reveal gas mixing, migration, and contamination pathways. Typical thermogenic ethane and propane values average ~-30‰ (range ≈-60‰ to -16‰), while abiotic sources can be comparatively enriched (sometimes approaching -5‰), and microbial systems are characterized by the absence or trace-only levels of these gases. By analyzing methane, ethane and propane together, SGS Beta delivers compound-specific isotopic fingerprints that provide a robust framework for source attribution, environmental monitoring, and petroleum system studies.
The δ13C can suggest formation pathways; however, combining it with 14C provides both process and age information, creating a powerful dual-isotope framework. By integrating δ13C analysis with radiocarbon (14C) measurements it is possible to determine the 14C activity (or age), origin, and fate of hydrocarbon and associated atmospheric gases (CO and CO2). Samples can be analyzed and reported using either Biogenic Hydrocarbon Gas 14C Testing via ASTM D6866 or submitted for radiocarbon analysis for reporting of Measured pMC (MpMC), conventional pMC (CpMC), apparent radiocarbon age in BP, and δ13C.
Contact your Account Manager or lab@radiocarbon.com to receive a Compound Specific Isotope Analysis questionnaire, which must be completed, submitted, and approved by our laboratory team prior to sample submission. Gas chromatography analysis using ASTM D7833 can also be requested alongside our isotopic testing services for methane, ethane, propane, and total carbon (bulk gas) samples.
Sample Volume and Container Requirements: 2% (v/v) in a 0.6 L multilayered aluminum foil gas bag or pressurized IsoTube.
Read our full guidelines for gas samples submission here.
For questions and more information, please contact us at lab@radiocarbon.com.
References
Page last updated: April 2026